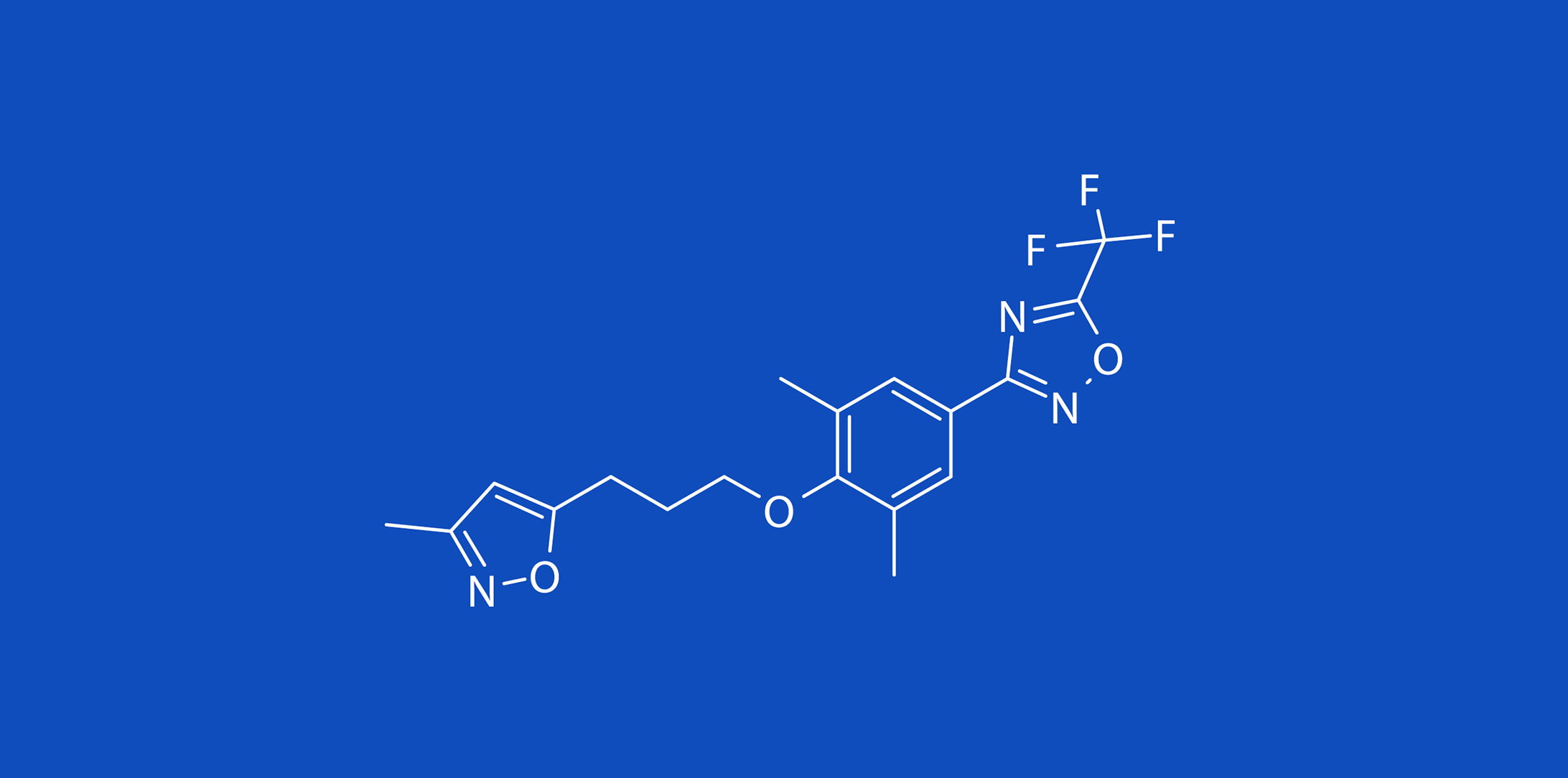
Pleconaril
Pleconaril is a direct acting antiviral (DAA) that is mainly effective on the rhinovirus subgroup of EV. The drug was developed in the 1990s for treating the common cold and exacerbations of asthma.
Pleconaril exerts its effects by acting directly on the virus, interfering with several processes in the virus’s replication. In 2001, Viropharma, Inc. submitted a new drug application to the FDA for use of pleconaril in the treatment of the common cold. The application was rejected based on safety concerns mainly attributable to the risk of serious drug interaction due to its effects on the CYP3A4. Pleconaril has also been used in several compassionate use programs for the treatment of serious EV infections.
Together with Oslo University Hospital, Curovir (through its sister-company Apodemus) is evaluating pleconaril given in combination with ribavirin in children with newly diagnosed type 1 diabetes (https://doi.org/10.2337/db14-1370). This trial is sponsored by Oslo University Hospital and Curovir (via Apodemus) provides the drugs in the trial and owns certain rights to the results. In addition, Curovir (through Apodemus) previously evaluated pleconaril for use in Alzheimer’s Disease (https://doi.org/10.3233/ADR-210301).